Establishment Registration
https://www.fda.gov/medical-devices/how-study-and-market-your-device/device-registration-and-listing
Any establishment that is involved in the production and distribution of a medical device intended for use in the United States is required to register annually with the FDA. The establishment must be registered at least 30 days before it can legally begin exporting the device into the United States. Depending on the device classification, it may be exempt from premarket approval or require premarket clearance or approval. The device cannot be imported until the approval has been granted.
Initial Registration
The establishment can submit an Initial Registration at any time during the calendar year. However all registrations expire at the end of the U.S. Fiscal year, which is September 30th. The annual registration fee must be paid prior to the initial registration and applies from that point until the end of the fiscal year.
Annual Renewal
Even if there is no change to the registration or listing information, the registration must be renewed between October 1st and December 31st for the next year. The Annual registration fee must be paid prior to submitting the registration renewal information.
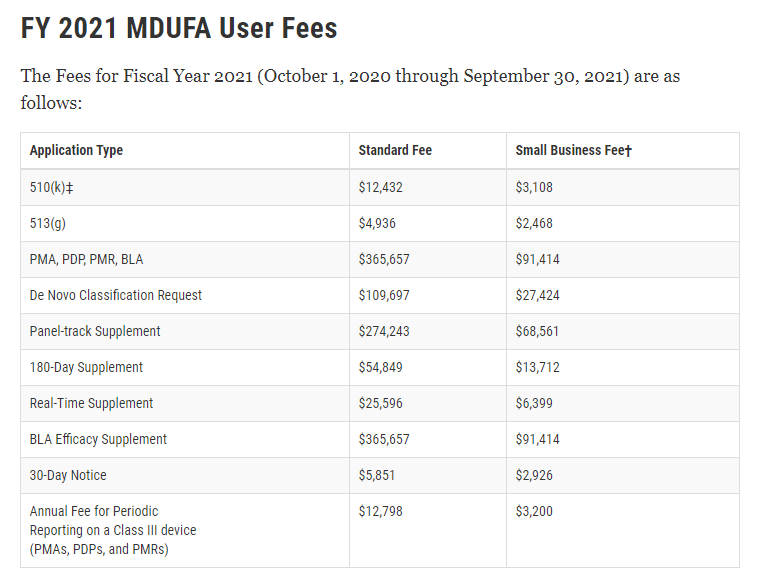
User Fees
Medical device companies pay user fees at initial registration and listing, annual renewal and for premarket notifications. The medical device user fees can be found here: and are summarized below for FY 2021.
The Annual Establishment Registration User Fee changes each year and must be paid each year between October 1st and December 31st to maintain your establishment registration for the next year.
| Year | FY 2021 |
|---|---|
| Fee | $5,546 |
Note:
If your company finds itself registering with the FDA late in the summer months, with your final registration approval coming in late summer, you will still have to renew your registration between October 1st and December 31st and pay the Annual Registration fee again. If possible, delay your initial registration until after October 1st.







